Models :Plane Of Symmetry
1. Benzene 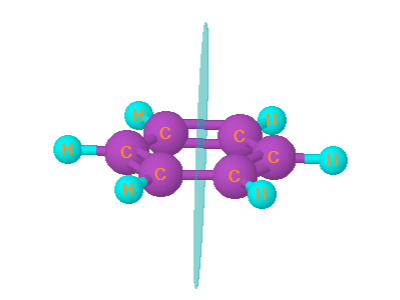

Benzene Link
Benzene is palnar. It has 7 plane of symmetry.
2. Methane 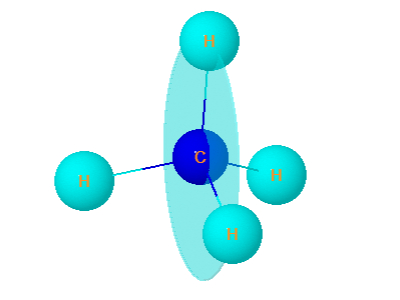

Methane Link
Methane is optically inactive . It has plane of symmetry .
3. Ethene 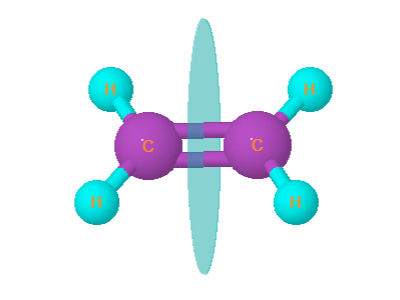

Ethene Link
Ethene is planar. It has plane of symmetry . Ethene is optically inactive .
4. Ethyne 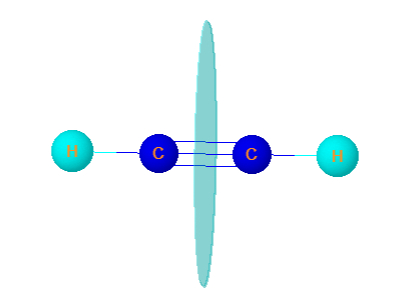

Ethyne Link
Ethyne is linear. It has infinite plane of symmetry. It is optically inactive.
5. Dichloromethane 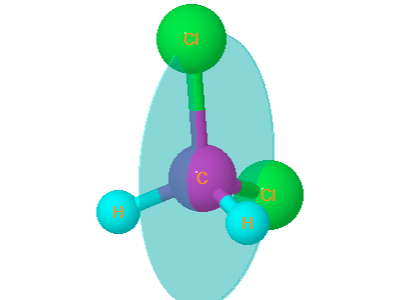

Dichloromethane Link
Dichloromethane is tetrahedral . It has plane of symmetry passing through both chlorine atoms or through both H atoms. It is optically inactive.
6. Chloroform 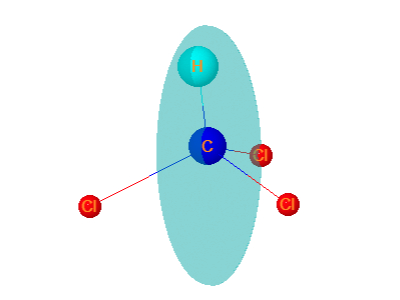

Chloroform Link
Chloroform is tetrahedral.It has plane of symmetry passing through H and one of Cl atom.
7. Cyclopropane 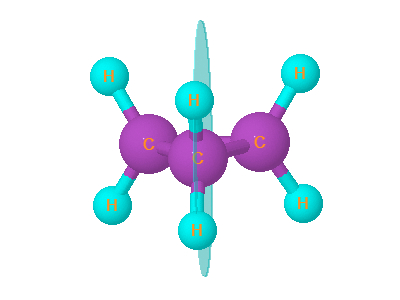

Cyclopropane Link
Cyclopropane is planar. It has plane of symmetry . It has 4 plane of symmetry.
8. Z12dichlorocyclopropane 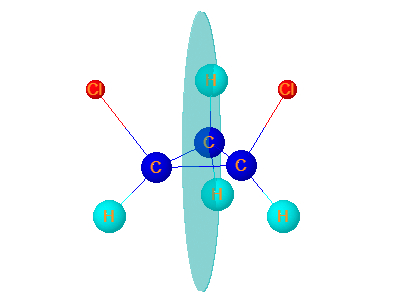

Z12dichlorocyclopropane Link
Z-1,2-dichlorocyclopropane has one plane of symmetry.It is symmetrical hence optically Inactive.
9. E12dichlorocyclopropane 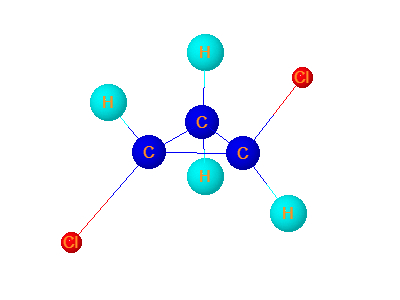

E12dichlorocyclopropane Link
E-1,2-dichlorocyclopropane has no plane of symmetry. Mirror image of E-1,2-dichlorocyclopropane is not superimposable on it. It is chiral and optically active.
10. Bromochloromethane 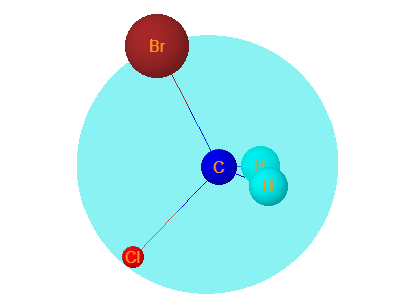

Bromochloromethane Link
Bromochloromethane has one plane of symmetry.It is symmetrical hence optically Inactive.
11. Bromochlorofluoromethane 

Bromochlorofluoromethane Link
Bromochlorofluoromethane has no plane of symmetry. Mirror image of Bromochlorofluoromethane is not superimposable on it. It is chiral and optically active.
12. 2-Butanol